The main indication for idarucizumab was reversal of anticoagulation for persistent bleeding (46.4%), followed by surgery (44.9%). Patient characteristics, concomitant conditions and laboratory parameters are reviewed in Table 1. Median aPTT was 45.6 seconds and was prolonged in 72.1% (49). The mean age was 73.5 ± 13.9 years, and 55.6% were men. Ninety-six percent received dabigatran for prevention of stroke and embolism in nonvalvular atrial fibrillation and 4% received it for the treatment of thromboembolic disease. Results: A total of 69 patients from 8 hospitals in Madrid were included. Cumulative survival was calculated by dividing the number of patients alive by the number of patients in each indication category for idarucizumab in a 30-day post-infusion period. 
Quantitative data are presented as mean ± standard deviation (SD) or median (interquartile range -IQR-). Qualitative data are presented as frequencies and percentages. 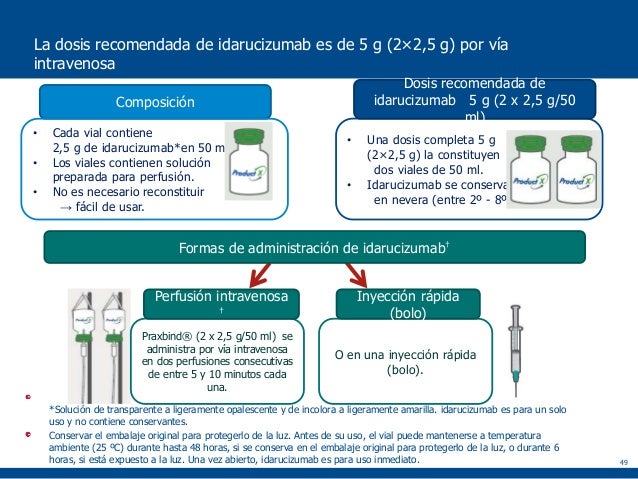
Demographic information, comorbidities, laboratory parameters, dabigatran indication, anticoagulation resumption, adverse events related to idarucizumab and death within 30 days were collected from medical records. Methods: Patients with electronic prescription of idarucizumab between June 2016 and July 2021 were included. The aim of the study was to describe the actual experience with idarucizumab in different centers in Madrid. It has been available in Spain since June 2016 and is indicated for imminent surgery or invasive procedures and life-threatening bleeding. Introduction: Idarucizumab is a humanized monoclonal antibody fragment that binds to dabigatran and reverses its anticoagulant activity. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
